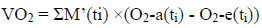All published articles of this journal are available on ScienceDirect.
Development of a Breath Analyzer for O2 and CO2 Measurement
Abstract
Background:
Breath analysis can be used to screen disease and detect physical conditions. Many research studies have investigated various sensors or techniques that were used to detect gas concentrations and temperature. However, the studies were less concerned about the performance of sensors.
Methods:
In this study, we developed and tested a portable breath analyzer for exhaled gas measurement. We detected air flow rate, temperature, humidity, CO2 and O2 concentrations. Commercial sensing elements were used to assemble this breath analyzer. All sensors were calibrated to ensure their accuracy. The response times of different sensors were considered. We analyzed breath during three states; 1) seating and resting for 5 min, 2) walking for 5 min and 3) running for 5 min in test participants. Fifteen students 20 to 34 years of age were the test participants.
Results:
Heat, humidity and CO2 production and O2 consumption were calculated from these measured data and were used to assess the performance of the analyzer. The Respiratory Quotient (RQ) were estimated by the ratio of VCO2 to VO2. The RQ values for resting, walking and running states were 0.899, 0.865 and 0.785, respectively. Walking and running significantly increased the heat and humidity production of the human body, corresponding to the human physiological function.
Conclusion:
The RQ value is usually in the range of 0.8 to 0.85. The measurement results for our exhaled analyzer were close to this range. From the respiratory quotient values, the performance of this breath analyzer was reasonable.
1. INTRODUCTION
Breath analysis is an in-situ and non-invasive method to observe the human body function [1]. It can be used to screen disease and detect physical conditions. Many devices and techniques have been developed to perform breath analysis [2, 3]. The exhaled breath is nearly saturated (100% relative humidity, RH) and is at a higher temperature than in normal air. Oxygen consumption (VO2) and carbon dioxide production (VCO2) are important variables for analyzing health conditions. The humidifying performance provides information about the physical conditions of the respiratory system. Devices to measure temperature, humility and other gas concentrations of exhaled air need to be developed [3].
Popov et al. [4] reported an individual-use device to measure Exhaled Breath Temperature (EBT) and found that the EBT was higher for test participants with asthma than controls. A thermal chamber was used to eliminate the effect of surrounding conditions, and the resistive thermometer was selected. The authors then constructed the second and third type of devices for EBT measurement [5, 6]. Then, the requirement of the portable EBT and the utilization of this device to detect a pathological process in the respiratory system were introduced in detail [7]. The factors such as ambient temperature affecting the EBT measurement were investigated and the application of EBT for respiratory diseases was discussed.
Logie et al. [8] measured EBT in 60 healthy children and found that room temperature, total lung capacity and slow vital capacity affect EBT. Bade et al. [9] used an X-Hall device to compare EBT between healthy people and patients with cystic fibrosis and reported that two groups have similar plateau temperatures. However, these patients had a slower increase rate with EBT.
Scheenstra et al. [10] studied intra- and inter- patient variability in endotracheal temperature and humidity and observed their heat and moisture exchange. The temperature was detected by thermocouple wires and humidity was measured by the Vaisala transmitter. The calibrations of temperature and humidity sensors were described by Zurr et al. [11].
Niesters et al. [12] evaluated the performance of the RespiR8 monitor to measure exhaled humidity. The humidity sensing element was made by the two electrodes on a polymer surface. However, the authors did not mention the effect of response time on sensors.
Wernecke and Wernecke [13] introduced humidity measurement for medical application and recommended capacitive and resistive humility probes. The problems with these electronic sensors were response time, condensation on the sensor at high humidity and contamination. The psychrometric measurement was the other choice. However, the air velocity passing the wet-bulb probe was the limiting factor. Folke et al. [14] assessed the performance of airflow, temperature, humidity and CO2 sensors with clinical aspects. They found that adequate devices were still lacking and miniature CO2 sensors needed to be developed. Breen [15] reported the effect of temperature and humidity measurement on the O2 uptake per breath during anesthesia by the validation of a pulmonary O2 uptake model. Rosenbaum [16] developed a new clinical bymixer to detect temperature, humidity, O2 and CO2 concentrations in exhaled air. Dry and wet temperatures were measured by thermocouple wires. However, the authors did not mention the sensing element of O2 and CO2. Szalados et al. [17] described a technique for estimating pulmonary function by calculating gas flow rate, O2 and CO2 concentrations. Spirometry was used to measure the gas flow rate. A fast paramagnetic sensor was used to detect O2 concentration. CO2 concentration was measured by an infrared sensor. They found that the CO2 data were not as sensitive as the O2 data, that is, the CO2 data were less responsive to physiologic charges [17]. Hunter et al. [18] described a smart breath diagnostic system with O2 and CO2 concentrations as the core sensing technique. The CO2 concentration was detected by a Near-Infrared Ray (NIR) CO2 analyzer. However, the sensing element of the O2 concentration was not reported. Smallwood et al. [19] compared the performance of two breath-by-breath devices. They mentioned that the O2 and CO2 sensors have been calibrated with a multipoint gas calibration method.
Vincent et al. [19] developed a portable breath analyzer to measure temperature, RH, flow-rate, O2, CO2 and Carbon monoxide (CO) concentration. CO2 was detected by a non-dispersive infrared CO2 sensor and O2 was measured by an electrochemical O2 sensor. They found that the response time of CO2 and O2 concentrations was too slow for breath-by-breath analysis [20].
The quantity of heat, humidity, CO2 production and O2 consumption of exhaled breath air could provide useful clinical information [15, 16, 21]. Many investigators mentioned various sensors or equipment used for determining gas concentrations and temperature. However, the performance of these sensors was less concerned. Temperature and CO2 sensors are physical sensors and humidity and O2 sensors are chemical sensors. The filter of the sensors’ enclosure is used to protect these sensing elements. However, the filter function may delay the reaction time. The calculation of medical parameters involves the multiple flow rate value and other measurement values. The required precise time and frequency of matching flow and gas concentration signals are required to collect useful information.
In this study, we developed and tested a portable breath analyzer for O2 and CO2 measurement. This breath analyzer was composed by these commercial sensors. The effect of the response time of sensors was observed. Heat, humidity and CO2 production and O2 consumption were calculated to assess the performance of this analyzer.
2. MATERIALS AND METHODS
2.1. Breath Analyzer
The diagram of the exhaled breath analyzer is in Fig. (1). The outside air is inhaled into the nose and then exhaled from the mouth, then air passes the filter and flows into the breath analyzer. The antibacterial filter (Cosmed, Rome, Italy) was used as the mouthpiece to prevent cross-contamination of bacteria. The temperature, relative humidity, CO2 and O2 concentrations of ambient air were measured using the same types of sensors. The selection of the mouthpiece was according to the study of Prabhakar et al. [2] without introducing substantial back pressure.
To avoid the effect of ambient temperature, the breath analyzer was surrounded by insulation materials. The flow rates of inhaled and exhaled air were measured and the change was small. It indicated that the flow resistance was small.
2.2. Experimental Devices
The specification of sensors is in Table 1.
K-type thermocouple wires (Omega Engineering, Connecticut, USA) were used to measure air temperature. The diameter of the wires was 0.15 mm and the response time < 0.05 s. To ensure the accuracy of temperate measurement, thermocouple wires were calibrated by a temperature calibrator (TC-2000, Instructed As, Bekkeveien, Norway). After calibration, the accuracy of temperature sensors was within 0.1°C.
The Shinyei THT-B7T transmitter (Shinyei Kaisha Technology, Kobe, Japan) was used to measure RH. The sensing probe is the macromolecule element. The accuracy of this humidity sensor was within 0.7% RH after calibration.
The Vaisala GMP20 transmitter (Vaisala, Helsinki, Finland) was used to measure CO2 concentration. The device was composed of the infrared ray probe, light filter, a light detector, gas diffusive membrane and pumping tube. The sensing device was calibrated by an SGD 710C gas divider (STEC Inc. Kyoto, Japan). The CO2 concentrations for calibration were adjusted by mixing the ratio of the pure CO2 and N2 gas. The accuracy of the CO2 sensor was within 50 ppm after calibration [22].
| - | Temperature Sensor | RH Sensor | CO2 Sensor | O2 Sensor |
|---|---|---|---|---|
| Sensing Element | K-type Thermocouple |
Macro-molecule HPR-MQ | Infrared Ray | Galvanic Type |
| Measuring range | 0 to 50°C | 20% to 90% RH | 0-2% | 0-100% |
| Accuracy before calibrating | 0.5°C | 3% RH at 25°C | 400ppm | 0.2% |
| Precision | 0.1°C | 1.0% RH | 10ppm | 0.01% |
| Accuracy after calibrating | 0.15°C | 1.1% RH | 50ppm | 0.05% |
The PSR 11-21 Oxygen sensor (Analytical industries Inc., CA, USA) was used to detect O2 concentration. This O2 sensor was calibrated by a two-point calibration technique. One point is the pure N2 gas for 0% O2 concentration and the other point is the O2 concentration in the air. The accuracy of the O2 sensor was within 0.05% after calibration.

The flow rate was measured by a Top-Trak 8255 flow meter (Sierra Instrument, CA, USA). The device was calibrated by its manufactures and the accuracy was 1.5% according to the manufacturer’s specifications.
2.3. Data Logger
All signals from sensors were connected to a dataTaker DT80 data logger (Thermos Fisher Scientific., Scoresby, Australia). The signals were calculated by their calibration equations and were then recorded. The sampling interval was ls.
2.4. Test Participants
Fifteen undergraduate and graduate students served as test participants (10 males and 5 females, age range 20 to 34 years). All participants were in a healthy state according to the regular physical check. All exhaled breath values were measured at 1-min periods. The physiological state before exhaled breath analysis was 1) seating and resting for 5 min, 2) walking for 5 min and 3) running for 5 min. During the measurements period, the room temperature and RH were maintained at 25 ± 3°C and 50 ± 15%. Three replicates were performed for each participant during the three states.
2.5. Data Analysis
2.5.1. Calculation of Humidity
The humidity of exhaled and inhaled air was calculated as follows [23]:
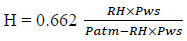 |
(1) |
where RH is relative humidity in decimals detected by the THT-B7T transmitter, Pws is the saturated air pressure in kPa and Patm is the air pressure in kPa.
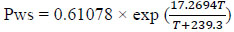 |
(2) |
where T is the temperature in °C.
2.5.2. The Production of Heat, Humidity and CO2
The production of heat (Vheat), humidity (Vhumi) and CO2 (VCO2)
was calculated as follows:
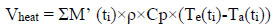 |
(3) |
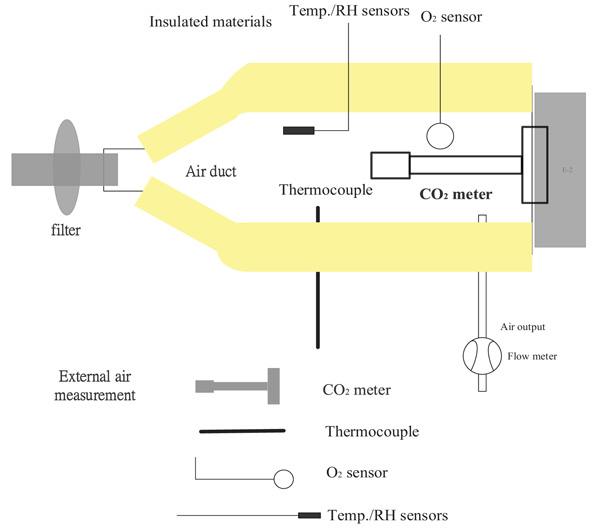
where M’ (ti) is the air flow rate in m3/s, ρ is the air density in Kg/m3, Cp is the specific heat in KJ/Kg, Te is the exhaled air temperature at ti in °C and Ta is the ambient air temperature at Ti in °C.
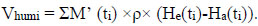 |
(4) |
where He(ti) is the absolute humidity of exhaled air at ti in Kg of H2O/Kg of air, and Ha(ti) is the absolute humidity of ambient air at Ti in Kg of H2O/Kg of air.
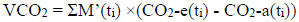 |
(5) |
where CO2-a(ti) is the CO2 concentration of ambient air at ti in ppm and CO2-e(ti) is the CO2 concentration of exhaled air at ti in ppm.
3. RESULTS AND DISCUSSION
3.1. Exhaled Gas Curves
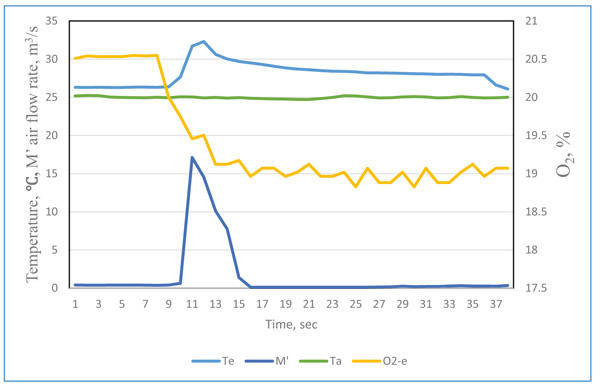
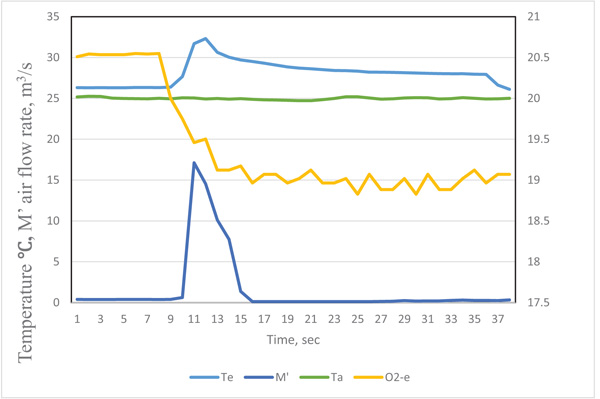
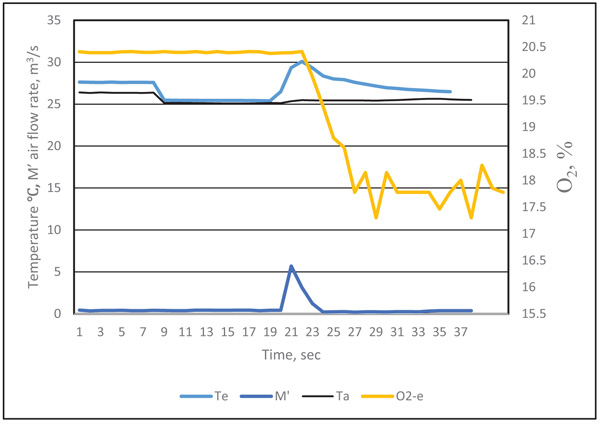
The original data recorded by the data logger at time i was labeled as τi .The typical curve for the quantity of exhaled breath gas of a test participant in the resting state is in Figs. (2 and 3), showing the effect of the response time of sensors on the measurement result. When the exhaled breath air entered into the analyzer, the air rate increased rapidity and the temperature signal had a similar trend. The air rate curve showed an abrupt increase in the quantity of air flow when the exhaled air passed the gas analyzer. The response of the exhale temperature showed a simultaneous increase in the temperature. The distribution of humidity, O2 and CO2 all revealed a delay in the response of 1.0, 3.0 and 3.0 s, respectively.
The inconsistent response could be attributed to the response time of sensors. The diameter of thermocouple wires was only 0.15 mm and its time constant was very small. The air passed the air rate meter by air pressure, the air rate was measured by very thin hot-wired temperature sensors, so the response time was fairly short. The humidity sensor had a 1 s delay because of its high molecule structure of sensing materials. The CO2 meter had a membrane to filter the contamination, and the membrane retarded the sampling air entering the detected cell. The gas diffusion membrane and the reaction of electrodes of the O2 probe also delayed the response time.
We considered the effect of the response time for different sensors. The revised exhaled gas curves were adjusted by their delay periods. To obtain precise timing of the measured data, the precision matching of signals were as follows: the temperate Te(ti) = Te(τi), air flow rate M’e(ti) = M’e(τi), humidity He(ti) = He(τi+1), CO2 concentration CO2-e (ti) = CO2-e (τi+3), and O2 concentration O2-e (ti) = O2-e(τi+3). By this time adjustment, the measurement of air flow rate, temperature and gas concentrations could be matched.
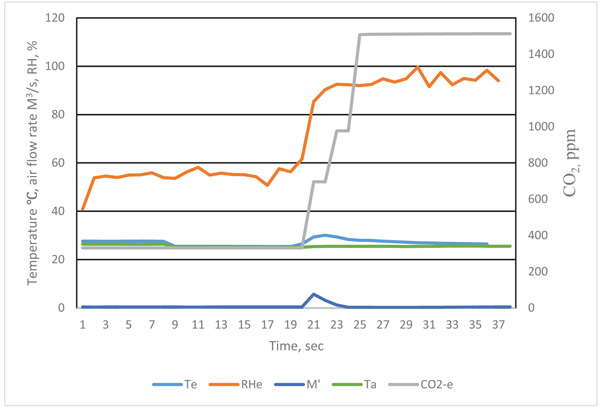
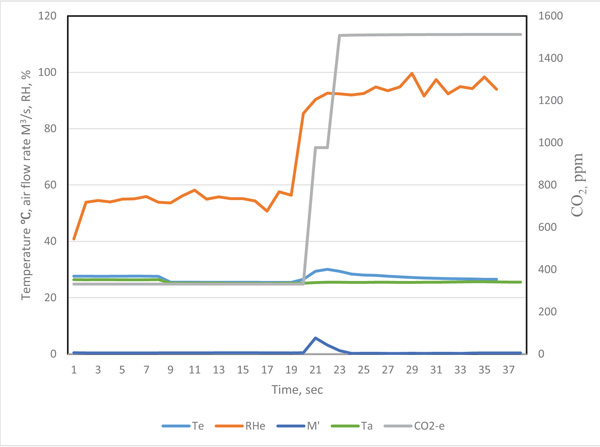
The revised curves are presented in Figs. (4 and 5). The distribution curves of temperature, air flow rate and gas concentrations were more reasonable. The actual respiration curves are shown in Figs. (4 and 5).
The distribution curves for the quantity of exhaled breach gas for a test participant after walking 5 min are in Figs. (6 and 7) and also show inconsistent results.
The revised curves for the exhaled breath gas are in Figs. (8 and 9). The results were reasonable for exhaled gas measurement.
The revised curves for the exhaled breath gas are after 5 minutes’ running in Figs. (10 and 11). The maximum CO2 concentration was near 2%. These results were reasonable for exhaled gas measurement after heavy exercise.
The diversity of Figs. (4, 5, 8-11) indicate that temperature, humidity and gas concentration patterns are affected by complex physiological factors.

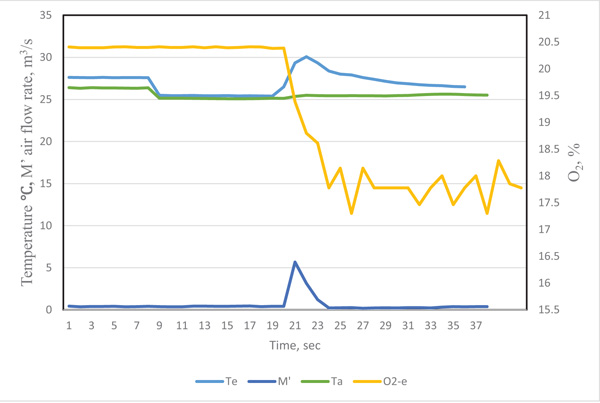
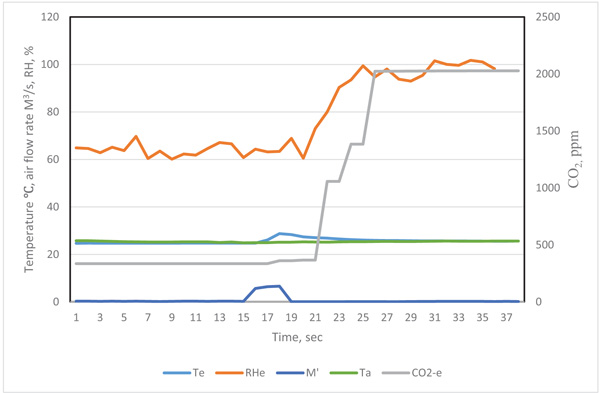
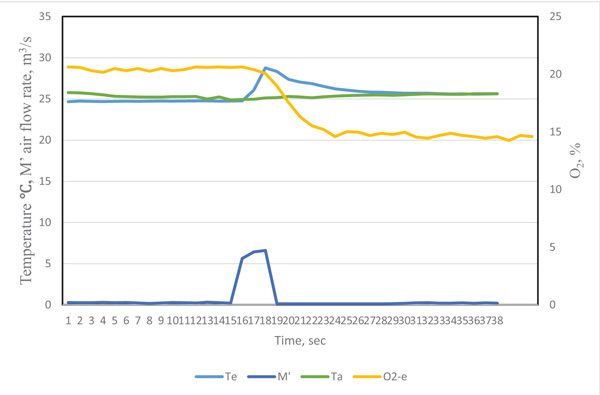
| Conditions | Heat (KJ) |
Humidity Kg of H2O |
CO2 mg |
O2 mg |
|---|---|---|---|---|
| Rest | 165.7(22.5)* | 0.41(0.10) | 46.3(9.0) | 51.5(9.8) |
| Slight exercise(SE) | 259.2(41.7) | 0.64(0.14) | 80.6(15.5) | 93.1(21.0) |
| Heavy exercise(HE) | 291.7(58.7) | 0.74(0.16) | 90.8(18.7) | 115.7(25.4) |
| SE/Rest | 1.56(0.31) | 1.56(0.30) | 1.74(0.34) | 1.81(0.33) |
| HE/Rest | 1.76(0.38) | 1.81(0.37) | 1.96(0.36) | 2.25(0.41) |
3.2. Physiological parameters
The physiological parameters of heat, humidity and CO2 production and O2 consumption calculated by Eqs. (3-6) are in Table 2.
The average heat production for resting, walking and running states was165.7 ± 22.5, 259.2 ± 41.7 and 291.7± 58.7 kJ, respectively. The walking and running state significantly increased the heat production of the human body. The results correspond to the physiological function.
Humidity production of three states was 0.41 ± 0.10, 0.64 ± 0.14 and 0.74 ± 0.16 kg, respectively. The walking and running state enhanced the increase in moisture during the exhale process. The mass of the CO2 production of three states was 46.3 ± 9.0, 80.6 ± 15.5 and 90.8 ± 18.7 mg, respectively. The mass of the O2 consumptions of three states was 51.5 ± 9.8, 93.1 ± 21.0 and 115.7 ± 25.4 mg, respectively. The higher values of the standard deviation for various parameters of three states indicated the diversity of sample conditions.
The Respiratory Quotient (RQ) is estimated by the ratio of VCO2 to VO2. RQ could be used in analysis and diagnosis of chronic obstructive pulmonary disease and liver disease. The RQ values for resting, walking and running states in this study were 0.899, 0.865 and 0.785, respectively. The RQ value is usually in the range of 0.8 to 0.85. The measurement results for our exhaled analyzer were close to this range. From the validation of physiological parameters calculated from the measurement values of this breath analyzer, its performance was reasonable.
Christopher et al. [24] investigated the stable performance of the K4b2 portable metabolic analyser in rest, walking and running conditions. The respiratory variables included O2 consumption and CO2 production. Goedecke et al. [25] studied variability in the respiratory exchange rate at rest and exercise at different levels. In trained athletes, the average RQ values for resting, 25% and 50% sustained peak power output (Wpeak) were 0.8, 0.84 and 0.89, respectively. The ratio was increased with increasing peak power output. Our RQ values were close to these results. The difference between the previous results and our finding may be explained by the participants. The Goedecke et al. study [25] was of trained athletes with mean age 31±7 years. In our study, all participants were undergraduate and graduate students with age range 20 to 30 years.
The calibration of sensors is important to ensure the accuracy of the measurement system. From a review of the literature, only the study of Scheenstra et al. [10]. and Swallwood et al. [19] mentioned the calibration of sensors. Hunter et al. [18] reviewed different sensors that could be used as smart sensor systems. However, the performance and calibration method of these sensors were not reported.
Folke et al. [14] introduced methods and devices for respiratory monitoring on temperature, humidity, carbon dioxide and air flow rate. They listed the problems of the sensors for respiratory measurement. In our study, five types of sensors were used to assemble a portable analyzer, the performance was reasonable. More data will be collected in the further study.
Popov et al. [4, 6] adopted a resistive thermometer to measure EBT. The advantages of this resistive sensing element are reliability and accuracy. However, the longer response time was its disadvantage. Popov et al. [5] proposed a first-order system response equation to eliminate the response time problem. In our study, we adopted very thin thermocouple wires. The rapid response of the metal wires decreased the response time, for more accurate of the temperature measurement.
Vincent et al. [20] developed a breath analyzer for metabolic rate measurement. The data at the initial 20 s were used to fit an exponential rise to maximum model and the required time to reach the 90% saturated values was 14.1 s for the CO2 sensor. The breath-to-breath interval was almost 5 s. The authors suggested using the nonlinear response model to improve measurement. In our study, the response time to reach the saturated levels was 3 s for CO2 and O2 measurement. Recently, the advanced technology of sensors has enhanced their performance, so the exhaled breath analyzer developed in this study have better performance.
Breen [15] developed a useful model with VO2 and VCO2 values to calculate the metabolism of a patient during anesthesia. The important parameters of these models were air flow rate, temperature, humidity, O2 and CO2 concentrations. Roseubaum et al. [16] developed several equations to calculate some parameters from the air flow rate and gas concentrations to evaluate metabolic gas exchange in a critical care unit.
Preiss et al. [21] developed a model to calculate pulmonary blood flow by measuring O2 and CO2 concentrations. The technique developed in this study included the calibration and the matching of the response time of sensors. It could be used to measure these variables. The measured values of these sensors are then used as variables for these physiological models.
The use of the infrared measurement of carbon dioxide in the human breath in history was introduced in detail [26]. Nowadays, the development of microprocessor systems is rapid and the cost is decreasing. The signals of sensors of the portable breath analyzer we developed can be connected to a data-logger, then the delay in response time for different sensors can then be considered and the precise timing of signals could be matched. The health index can then be calculated by previously established models embedded in the flash ROM of the microcomputer. With the adequate sensors and microprocessor systems, a smart sensor of the model-based approach could be developed for analyzing breath in the clinic.
CONCLUSION
Breath analysis can be used to screen disease and detect physical conditions. In this study, we developed and tested a portable breath analyzer for exhaled gas measurement. Air flow rate, temperature, humidity, CO2 and O2 concentrations were detected. The commercial sensors were used for this breath analyzer. All sensors were calibrated to enhance its accuracy. The response times of different sensors were considered. Heat, humidity, CO2 production and O2 consumption were calculated from these measured data. These calculated parameters were used to assess the performance of the analyzer. From the respiratory quotient values, the performance was reasonable. The smart sensor of model-based approach could be developed for analyzing breath in the clinic with the adequate sensors and a microprocessor system.
AUTHORS’ CONTRIBUTIONS
Hsuan-YuChen drafted the proposal, executed the statistical analysis, interpreted the results and revised the manuscript. Chiachung Chen reviewed the proposal, performed some experiments, interpreted some results and criticized the manuscript and participated in itsrevision. All authors have read and approved the final manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals were used in the study. The reported experiments in accordance with the standards set forth in one of below national guidelines and regulations: The US Public Health Service's "Policy on Humane Care and Use of Laboratory Animals," and "Guide for the Care and Use of Laboratory Animals."
CONSENT FOR PUBLICATION
Informed consent was obtained from all the participants.
FUNDING
The study has been supported by Ministry of Science and Technology of the Republic of China under Contract No. MOST-106-2313- B-005-006.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
The authors would like to thank the Ministry of Science and Technology of the Republic of China for financially supporting this research.